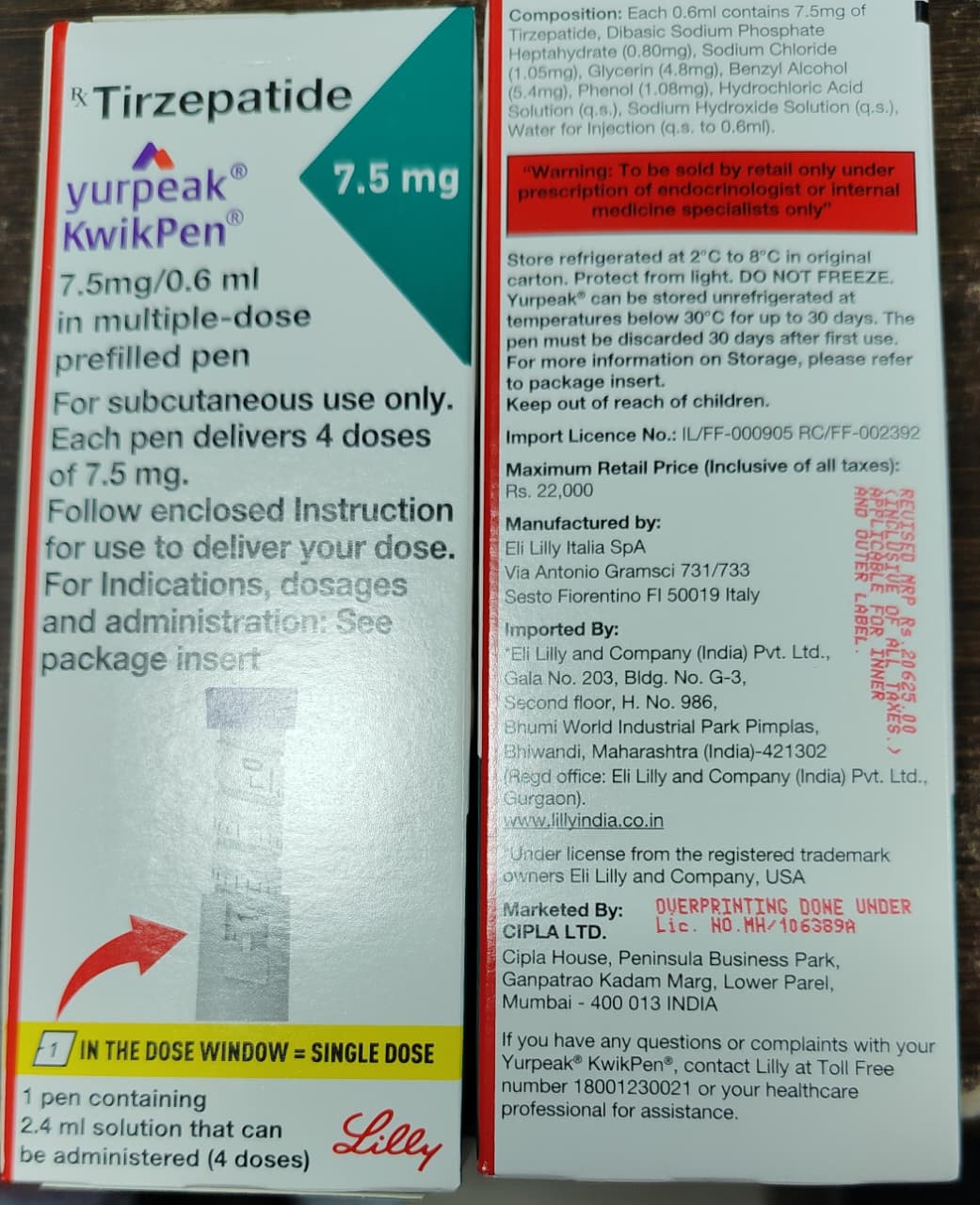
Yurpeak 7.5mg KwikPen
Yurpeak 7.5mg KwikPen
Details
Drug Name: Tirzepatide
Category: Antidiabetic / GLP-1 & GIP Dual Agonist / Injectable Therapy
International Brand: Yurpeak KwikPen
Available Strengths: 2.5mg, 5mg, 7.5mg & 10mg pre-filled pens
Product Description of Use:
KwikPen Tirzepatide 7.5mg is an advanced injectable diabetes treatment manufactured by Eli Lilly in Italy and marketed in India by Cipla. Developed as a once-weekly pre-filled injection pen, KwikPen Tirzepatide 7.5mg is designed to improve blood sugar control in adults with type 2 diabetes. With a convenient, ready-to-use device, KwikPen Tirzepatide 7.5mg supports long-term glucose management while also promoting weight reduction and metabolic improvement.
Uses:
• KwikPen Tirzepatide 7.5mg helps control elevated blood glucose in adults with type 2 diabetes
• Used alongside diet and exercise programs to support metabolic health
• Reduces risk of diabetes-associated complications
• May assist in weight reduction for improved treatment outcomes
• Useful for patients needing combination action on insulin sensitivity & appetite control
How it Works:
Tirzepatide, the active component in KwikPen Tirzepatide 7.5mg, acts on both GLP-1 and GIP receptors. This dual pathway enhances insulin production, suppresses excess glucose release from the liver, slows gastric emptying, and reduces appetite — resulting in efficient blood sugar control and improved weight management.
Dosage:
• KwikPen Tirzepatide 7.5mg is administered once weekly via subcutaneous injection
• The 7.5mg dose is typically used as the starting stage before gradual dose escalation
• Can be induced in the abdomen, thigh, or upper arm
• Use on the same day each week, at any time, with or without food
Side Effects:
• Nausea or vomiting
• Diarrhea or constipation
• Low appetite
• Abdominal pain
• Mild injection-site reactions
Precautions:
• Monitor the kidneys and liver functioning while and during treatment
• Not advised for type 1 diabetes or patients with diabetic ketoacidosis
• Avoid use in pregnancy or breastfeeding without medical direction
• Report severe gastrointestinal symptoms immediately
• Medical supervision essential for patients transitioning from insulin or other GLP-1 agents
Contraindications:
• Thyroid history of Personal or family
• Multiple endocrine neoplasia syndrome type 2
• Severe gastrointestinal disorders
• Known hypersensitivity to Tirzepatide
• Severe dehydration or vomiting
Interactions:
• May affect absorption of oral medications due to delayed gastric emptying
• Combined use with insulin or sulfonylureas may increase hypoglycemia risk
• Avoid combination with other GLP-1 agonists
• Caution with diuretics or medications affecting fluid balance